In recent years, a growing number of dryland producers have been exploring ways of producing their own nitrogen fertilizer, on-farm. For some, this means making their own biological products by brewing compost teas (opens in new tab) and making compost extracts that can be applied in a liquid form, for instance as a seed inoculant or foliar application (opens in new tab). Others are trying out a new technology sometimes called Plasma Assisted Nitrogen Fixation (PANF), which uses low-temperature plasma to fix atmospheric nitrogen into nitrate. For example, the company GreenLightning sells a system that installs on top of a liquid tote, and only requires water and 110V electricity to produce a nitrate solution (approximately 530 ppm nitrate-N, or 0.00442 lb N per gallon).

The idea of being able to self-produce nitrogen fertilizer, on-farm, is intriguing. But given how critical an input nitrogen is, why haven’t on-farm technologies emerged before now? To learn more, I spoke with Pascal Mickelson (opens in new tab), an applied physicist researching PANF for various applications. He described how electricity and plasmas are used to fix nitrogen and recent developments that could allow for more distributed nitrogen fertilizer production.
Electrification as an alternative to Haber-Bosch
Atmospheric N2 is a triple-bonded molecule and is famously difficult to break. The Haber-Bosch process, which has been used to produce almost all N fertilizer globally since World War II, applies high pressures and temperatures (400 to 500°C) to help break this bond. But as Mickelson put it, a thermal process like Haber-Bosch is non-selective. It puts energy into whole molecules and electrons, whereas only energized electrons are needed to break the triple bond. Plasmas create free electrons with significant kinetic energy and can break the N2 triple-bond at temperatures closer to 150-250°C. This selectivity, and the lower temperatures required, means plasma assisted fixation can be carried out with less energy input than Haber-Bosch.

Using electricity to fix N2 is not a new idea. In fact, the earliest industrial process for nitrogen fixation used electrical arcs (opens in new tab) to react N2 in the atmosphere with O2. In Norway, reactors were built in the 1910s alongside hydroelectric facilities that provided the necessary electricity. But it was the thermal process invented by the German scientist Haber, and scaled-up by Bosch, that was continually improved and refined, and became the preferred method globally. Today, ammonia production is highly centralized and highly efficient. There are estimated to be only about 450 ammonia plants world-wide (opens in new tab), producing the entire supply of synthetic nitrogen fertilizer.
Mickelson says that in the last 10 to 15 years there has been a renewed interested in electrifying nitrogen fixation. This is part of a trend towards electrifying industrial processes more generally. Electrification allows manufacturing to be powered with wind and solar, which not only reduces emissions, but can allow manufacturing plants to be built out more quickly, and in a decentralized way. This autonomy benefits industries, like farming, that are at the end of long supply chains. Most U.S. ammonia is co-produced with natural gas refineries in the Gulf Coast region, and the ammonia is subsequently processed into various forms of nitrogen fertilizer. Nitrogen fertilizers travel to the PNW about 2000 miles by rail and are then trucked to retailers and farms. This system is well-established and efficient. But the ability to make fertilizer locally, on-demand, would provide farmers a measure of independence and protection from supply-chain disruption.
Plasma-activated water
Mickelson points out that a limitation of Haber-Bosch is that it only produces ammonia. Plasma technologies, on the other hand, provide chemical paths to make a variety of nitrogen end-products. Plasma-activated water, in particular, provides a pathway to make nitrate (NO3–).
Richard Smith, a Washington-based supplier of GreenLightning, explained how their plasma-activated water system produces nitrate. An air compressor delivers ambient air to a gliding arc plasma. Essentially air is exposed to an electrical field, which energizes it and creates reactive gas molecules, including reactive nitrogen gas species (e.g. NO, NO2). The plasma-activated air is then brought into contact with water, which converts the reactive nitrogen gases to nitrate (NO3–) and nitrite (NO2–). The process also generates peroxide (H2O2). In fact, because peroxide has disinfecting and antimicrobial properties, it is a desirable component of plasma-activated water for uses such as seed treatments.
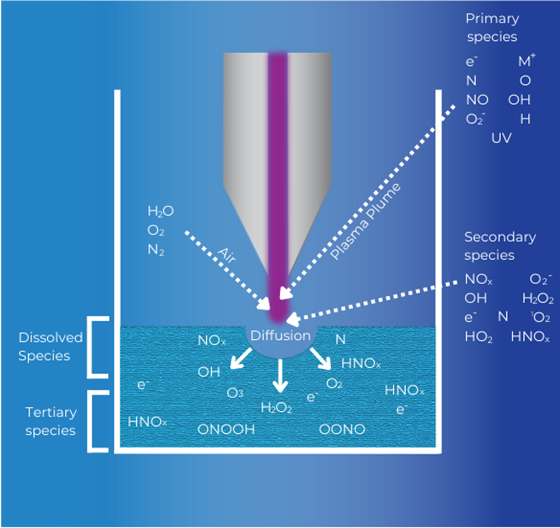
Mickelson explained that past research with plasma-activated water had difficulty producing sufficiently high nitrate concentrations, with sufficiently low energy input, to make the technology a strong candidate for commercialization. However, Smith explained that GreenLightning makes use of an injector that has improved interaction between reactive nitrogen species and water, and increased nitrate yield.
Smith says that the company is still improving their technology to increase the nitrate concentrations they can achieve and are partnering with farmers to conduct field testing. He says that GreenLightning fertilizer is well matched for fertigation systems, particularly as it does not contain salts as traditional nitrate fertilizers do. Some dryland farmers are also experimenting with GreenLightning fertilizer as a seed starter, and as a spring topdress and foliar fertilizer.
Farmers considering experimenting with foliar application should be aware that nitrate generally does not cross into leaves as readily as urea. Nitrate is a larger molecule and is negatively charged, and therefore does not cross waxy leaf cuticles effectively. However, there is evidence that foliar nitrate can improve wheat growth by acting as a regulatory signal, for instance by upregulating genes involves in starch synthesis (opens in new tab) and grain-filling. Research in horticultural crops has also looked at the antimicrobial properties of peroxide (which is most concentrated in freshly-prepared plasma-activated water, and declines over time). The peroxide in plasma-activated water is beneficial for disinfecting seeds (opens in new tab), activating plant defense responses (opens in new tab), and controlling foliar fungal disease (opens in new tab).
Other farm-scale nitrogen fertilizer technologies
Mickelson estimates that a few start-ups, perhaps numbering in the dozens, are exploring farm-scale nitrogen production using PANF. He says most are based in Europe, but California-based Nitricity (opens in new tab) received a fair amount of investor interest. Established in 2018, Nitricity built several versions of pilot plants (opens in new tab) using plasma-activated water to produce a liquid nitrate fertilizer. Ultimately the company shifted to developing organic fertilizers from waste plant materials, using electricity to power the combustion process. Smith noted that Nitricity has focused on developing fertilizer products, while GreenLightning has focused on providing nitrogen-fixation technology to farmers.
Other start-ups are exploring “green ammonia”, which generally refers to ammonia that is produced from N2 and H2, like Haber-Bosch, but with fewer emissions. Various technologies are used for green ammonia, but they often involve replacing some of the fossil energy (opens in new tab) required for Haber-Bosch with renewable energy sources. One green ammonia approach involves an electrochemical reactor (opens in new tab) (similar to a battery) to drive N fixation with less energy input.
Collectively, these innovations may provide PNW farmers more sources for nitrogen fertilizer in the coming decades. The products themselves, from compost teas that are biologically-rich, to plasma-activated water which has anti-microbial properties, are quite varied. There will be plenty to learn to efficiently and effectively operate on-farm production systems, store the products, and apply them to crops.
Contributed by Claire Phillips, WSU Research Associate with the Northwest Sustainable Agroecosystem Research Unit, USDA-ARS.
