Contributed by Joshua Miranda and Marcelo Moretti, Oregon State University
A widespread evolution of herbicide resistance
Annual bluegrass (Poa annua L.) is a winter annual species that thrives in cool, disturbed environments, including orchard floors, turfgrass, and rights-of-way. It is one of the most adaptable and persistent grass weeds in temperate agroecosystems. In Oregon hazelnut orchards, annual bluegrass has traditionally been controlled using a limited set of preemergence and postemergence herbicides, most commonly glyphosate, clethodim, paraquat, and pendimethalin. However, in recent years, hazelnut growers and crop consultants in the Willamette Valley have reported declining herbicide performance despite the use of full recommended label rates and proper application timings. These observations raised concerns about potential herbicide resistance.
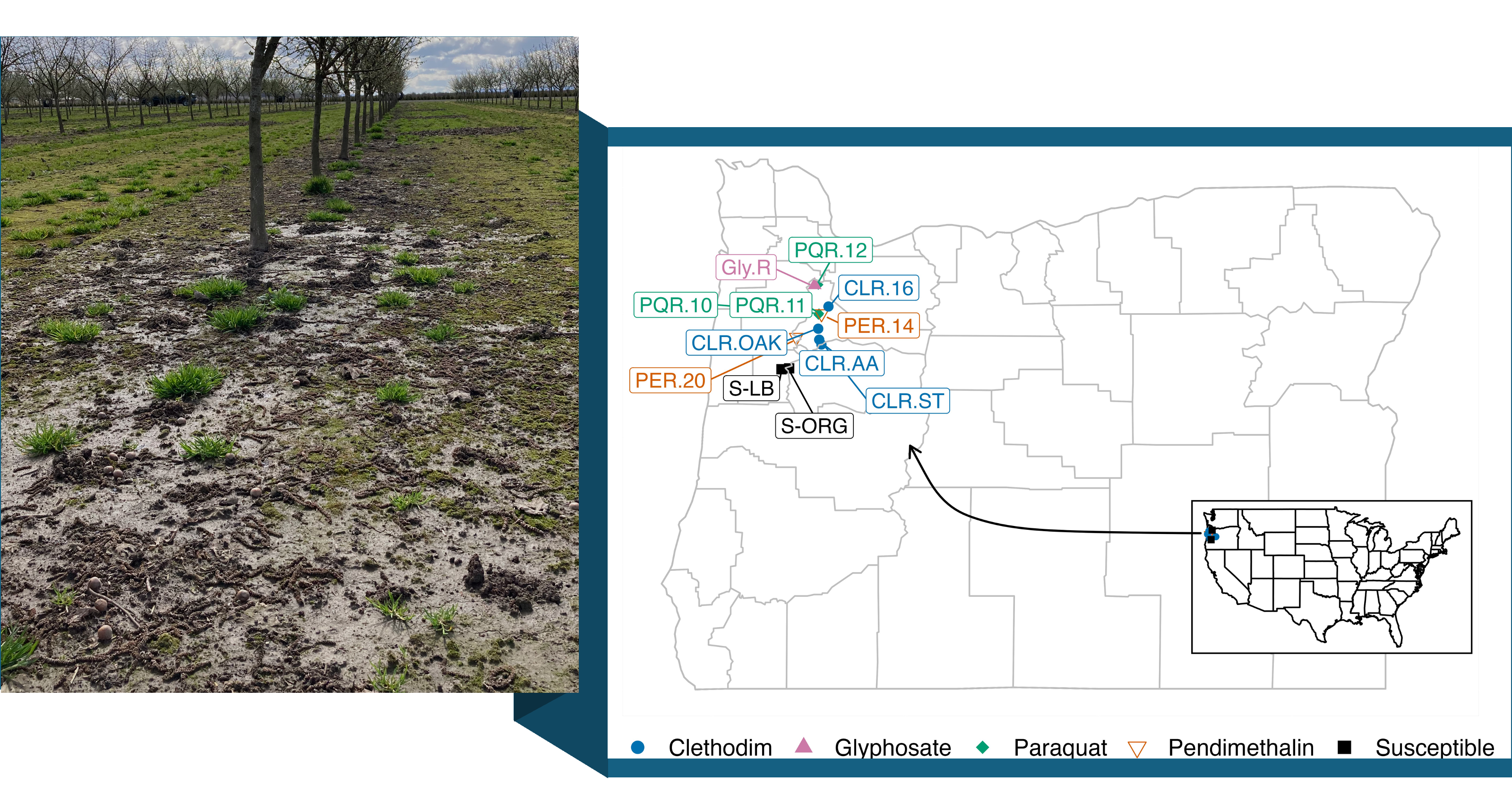
To investigate this issue, we collected annual bluegrass plants from hazelnut orchards where herbicides had failed (Fig. 1). A total of ten suspected resistant accessions were collected: one for glyphosate (Gly.R), four for clethodim (CLR.OAK, CLR.ST, CLR.16, CLR.AA), three for paraquat (PQR.10, PQR.11, PQR.12), and two for pendimethalin (PER.14, PER.20). Two susceptible accessions, S-ORG and S-LB, were included for comparison. S-ORG was collected from an organically managed orchard and S-LB from a turfgrass area. Each accession was screened for the herbicide it was suspected of resistance, using both seed-based assays and whole-plant dose-response bioassays under controlled conditions. Our results confirmed that all collected accessions were resistant to the herbicides in question. This is the first time resistance to clethodim has been confirmed in annual bluegrass anywhere in the world. It’s also the first case of paraquat resistance in this species in the U.S. These results highlight the rapid evolution of herbicide resistance of annual bluegrass and the urgent need to diversify weed management strategies.
A seed-based assay for early detection
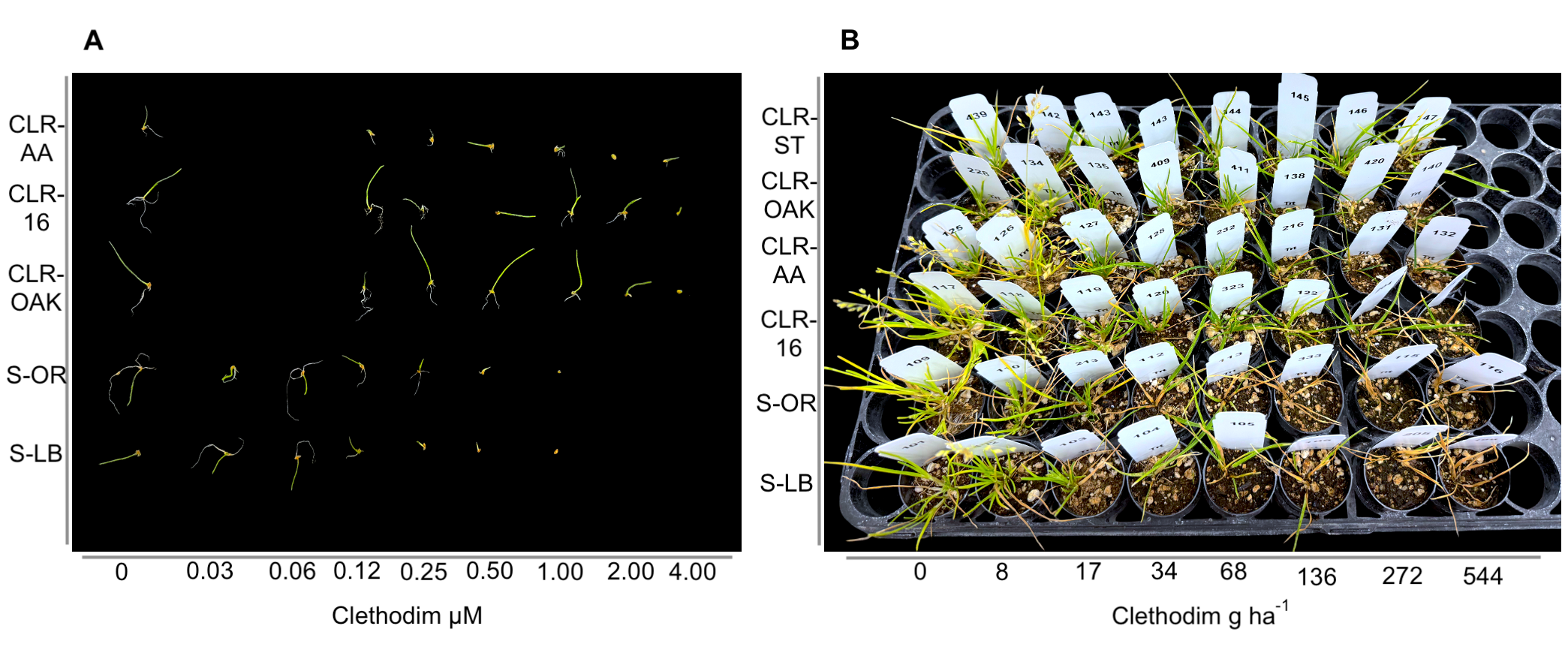
A major outcome of this research was the development and validation of a seed-based test that helps detect herbicide resistance early (Fig. 2). This method is simple, affordable, and scalable, offering a practical diagnostic tool for early detection of resistance. Seeds collected from suspected resistant plants are placed on moist filter paper inside sealed petri dishes. Four milliliters of herbicide solution are added, and the dishes are kept in a warm, illuminated area for 14 days. If the seeds germinate and grow despite the herbicide treatment, it may indicate resistance. Comparing results to a nontreated control and to a known susceptible seed sample helps confirm the diagnosis.
The test does not require specialized equipment and provides rapid and reliable results that are closely aligned with whole-plant bioassay outcomes. This test could prove invaluable for the early detection of resistance and allow growers to adapt their management strategies before resistant biotypes become dominant. The test offers a cost-effective alternative to traditional greenhouse trials and can complement molecular testing where available. It also supports high-throughput screening and has the potential for on-farm implementation. Our study demonstrated that seed-based results were consistent with whole-plant dose-response bioassays, confirming the method’s accuracy. Similar assays have been described for other weed species (Perez et al. 2021; Cutulle et al. 2009), and our findings show it can be successfully adapted for annual bluegrass.
The following concentrations were identified as discriminating concentrations, levels at which only resistant seedlings survived:
- Clethodim: 1 µM
- Pendimethalin: 12 µM
- Paraquat: 2 µM
- Glyphosate: 500 µM
These concentrations can serve as diagnostic markers for distinguishing resistant and susceptible phenotypes in field-collected populations, offering a practical framework for resistance monitoring.
What this means for Oregon growers
The emergence of herbicide-resistant annual bluegrass requires a shift from reactive to proactive strategies. Reliance on chemical control alone is unsustainable, particularly in systems with limited herbicide diversity. Effective resistance management must include rotation of herbicide modes of action, tank mixing with multiple effective sites of action, and integration of non-chemical strategies such as cultivation, cover cropping, mulching, and mowing. Regular scouting is essential to identify early escapes and prevent seedbank replenishment. The adoption of seed-based diagnostic tools can help growers make timely decisions, avoid ineffective applications, and preserve herbicide efficacy.
Takeaway
The confirmation of clethodim, pendimethalin, glyphosate, and paraquat resistance in annual bluegrass from Oregon hazelnut orchards reflects the adaptive potential of this species under herbicide selection pressure. This work establishes a baseline for herbicide resistance in annual bluegrass within Oregon hazelnuts. With integrated management, early detection tools, and improved stewardship, the industry can take meaningful steps to slow the spread of resistance and maintain effective weed control in the years ahead.
For assistance with herbicide resistance diagnostics or to learn more about integrated weed management strategies for hazelnut production, growers are encouraged to contact their local OSU Extension office or the Department of Horticulture at Oregon State University.
Acknowledgment
This work was supported by the Oregon Hazelnut Commission and Ferrero Hazelnut Company. We thank the growers who granted access to their orchards.
References
Cutulle MA, McElroy JS, Millwood RW, Sorochan JC, Stewart CN (2009) Selection of bioassay method influences detection of annual bluegrass resistance to mitotic‐inhibiting herbicides. Crop Sci 49:1088–1095
Perez MB, Beckie HJ, Cawthray GR, Goggin DE, Busi R (2021) Rapid On-Farm Testing of Resistance in Lolium rigidum to Key Pre- and Post-Emergence Herbicides. Plants 10:1879